A single electron is confined to a single-walled carbon nanotube (SWNT) of length
Hint: Use de Broglie's hypothesis (
ANSWER: The longest wavelength (ground) state of
a particle in a one-dimensional (1D) "box"
has ![]() and therefore (by de Broglie's hypothesis)
momentum
and therefore (by de Broglie's hypothesis)
momentum ![]() . For a "1D box" this large
the electron will be nonrelativistic (you can check this),
so we can set
. For a "1D box" this large
the electron will be nonrelativistic (you can check this),
so we can set ![]() and thus
and thus
![]() where
where ![]() is the time
for the electron to make a round trip to the other end
and back. Thus the electron transfers momentum
is the time
for the electron to make a round trip to the other end
and back. Thus the electron transfers momentum ![]() (by reversing its direction) to one end of the "string"
every
(by reversing its direction) to one end of the "string"
every ![]() , generating an average force
, generating an average force
![]() .
Then we just plug in
.
Then we just plug in ![]() m,
m,
![]() kg and
kg and
![]() J-s to get
J-s to get
![]() or
or
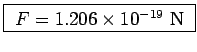 .
Since each end of the SWNT is being pushed "out"
by this force, it is the same as the tension
in the SWNT.1
.
Since each end of the SWNT is being pushed "out"
by this force, it is the same as the tension
in the SWNT.1
Making use of the EQUIPARTITION THEOREM, derive an equation analogous to the familiar 3D IDEAL GAS LAW (
ANSWER: The arguments developed for the time-averaged force
exerted on one wall of a cubical 3D container apply equally well
for the force acting on the boundary of a 1D "box" - namely,
for a single particle bouncing back and forth,
![]() ,
where
,
where ![]() is the length of the "box".
In this case there is only one direction of motion
(degree of freedom) so we can drop the
is the length of the "box".
In this case there is only one direction of motion
(degree of freedom) so we can drop the ![]() subscript on
subscript on ![]() .
The EQUIPARTITION THEOREM says that the thermal
average of the kinetic energy
.
The EQUIPARTITION THEOREM says that the thermal
average of the kinetic energy
![]() associated with
this translational degree of freedom is
associated with
this translational degree of freedom is
![]() , giving
, giving
![]() .
If we substitute this back into the formula for
.
If we substitute this back into the formula for
![]() we get
we get
![]() . For
. For ![]() particles all
doing this at the same time, we just multiply by
particles all
doing this at the same time, we just multiply by ![]() , giving
, giving
![]() or (ignoring the fact that the
actual force
or (ignoring the fact that the
actual force ![]() at any instant fluctuates minutely about the average
force
at any instant fluctuates minutely about the average
force
![]() )
)
![]() .
This is the 1D equivalent of the IDEAL GAS LAW.
Note that the only difference between this derivation and the one for
a 3D box is that here we didn't have to introduce the notion of
pressure as the force per unit area. This is simpler!
.
This is the 1D equivalent of the IDEAL GAS LAW.
Note that the only difference between this derivation and the one for
a 3D box is that here we didn't have to introduce the notion of
pressure as the force per unit area. This is simpler!
- What is the thermal speed distribution
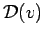 [in the textbook's notation,
[in the textbook's notation, 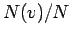 , as in Eq. (22-14) on p. 503]
for an ideal gas confined to a one-dimensional "box"?
It would be nice if you could find the right leading factors
(involving temperature and various constants)
to normalize the distribution so that
, as in Eq. (22-14) on p. 503]
for an ideal gas confined to a one-dimensional "box"?
It would be nice if you could find the right leading factors
(involving temperature and various constants)
to normalize the distribution so that
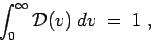
but I am mainly looking for its dependence on the speed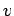 .
.
Hint: Again, use de Broglie's hypothesis and think of standing waves.ANSWER: Again the 1D case is much simpler than the 3D case because we don't have to worry about composing
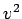 out of
out of
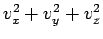 (or
(or 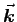 out of
out of 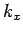 ,
, 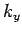 and
and 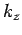 ).
There is just one direction, just one velocity component,
one momentum component and one wavelength to worry about
making commensurate with the length of the "box".
Thus the requirement that an integer
).
There is just one direction, just one velocity component,
one momentum component and one wavelength to worry about
making commensurate with the length of the "box".
Thus the requirement that an integer 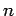 half-wavelengths
fit evenly into
half-wavelengths
fit evenly into 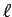 gives
gives
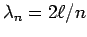 and so
and so
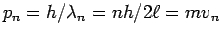 or
or
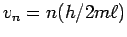 . Thus the possible values of
the speed
. Thus the possible values of
the speed 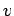 are evenly spaced every
are evenly spaced every 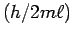 and the distribution of speeds varies only as the
Boltzmann factor
and the distribution of speeds varies only as the
Boltzmann factor
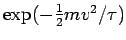 . This gives immediately
. This gives immediately
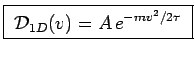 where
where 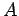 is a normalization constant that does not depend on
is a normalization constant that does not depend on 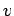 .
You get full credit for this result, but here's how to get
.
You get full credit for this result, but here's how to get 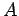 :
Let
:
Let
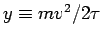 where
where 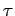 is treated as a constant.
Now,
is treated as a constant.
Now,
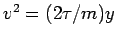 or
or
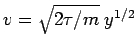 so
so
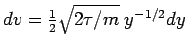 and we have
and we have
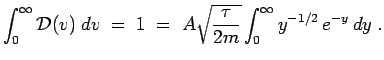 You can look up the definite integral; its value is
You can look up the definite integral; its value is 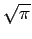 ,
giving
,
giving
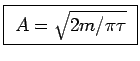 .
.
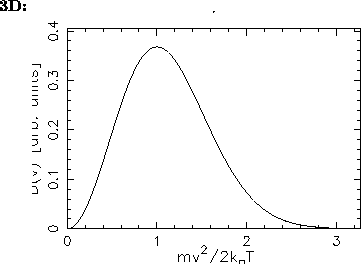
- Sketch this distribution for a given temperature
and compare its shape with that shown in the Figures
on p. 503 of the textbook.
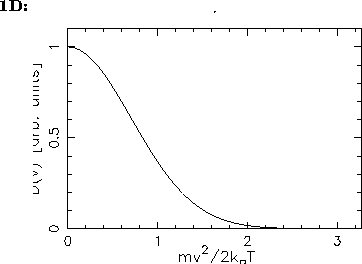
- What can you say about the most probable speed
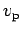 in the two different cases?
in the two different cases?
ANSWER: As stated in the textbook, the most probable speed for an ideal gas molecule in 3D is
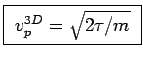 (i.e. when
(i.e. when
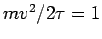 ).
For the 1D gas, however,
).
For the 1D gas, however,
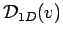 is missing that
extra factor of
is missing that
extra factor of 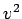 that forces its value to zero at
that forces its value to zero at 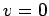 ,
so the resultant speed distribution has its maximum
at
,
so the resultant speed distribution has its maximum
at 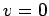 :
:
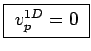 .
.